 
What two factors determine the strength of coulombic attraction? 37. Explain your answer to Question 34: Why does the atomic radius change? 36. Within a period, what happens to the atomic radius as the atomic number increases? 35. Explain your answer to Question 31: Why does the atomic radius change? He, Ne, Ar Li, Na, K, Rb, Cs, Fr Cs Br Ag THEY HAVE THE SAME NUMBER OF VALENCE ELECTRONS THE SIZE OF A NEUTRAL ATOM INCREASES ELEMENT BELOW HAS ONE MORE ENERGY LEVEL THAN ELEMENT ABOVEģ3. Within a group, what happens to the atomic radius as you go down the column? 32. What do we mean by the “atomic radius?” 31. Why do all the members of a group have similar properties? 30. What element is in the fifth period and the eleventh group? 29. Which halogen belongs to the fourth period? 28. Which alkali metal belongs to the sixth period? 27. List the three lightest members of the noble gases. s1 ALKALINE EARTH METALS HALOGENS NOBLE GASES ALKALINE EARTH METALS NOBLE GASES HALOGENS ALKALI METALSĢ4. 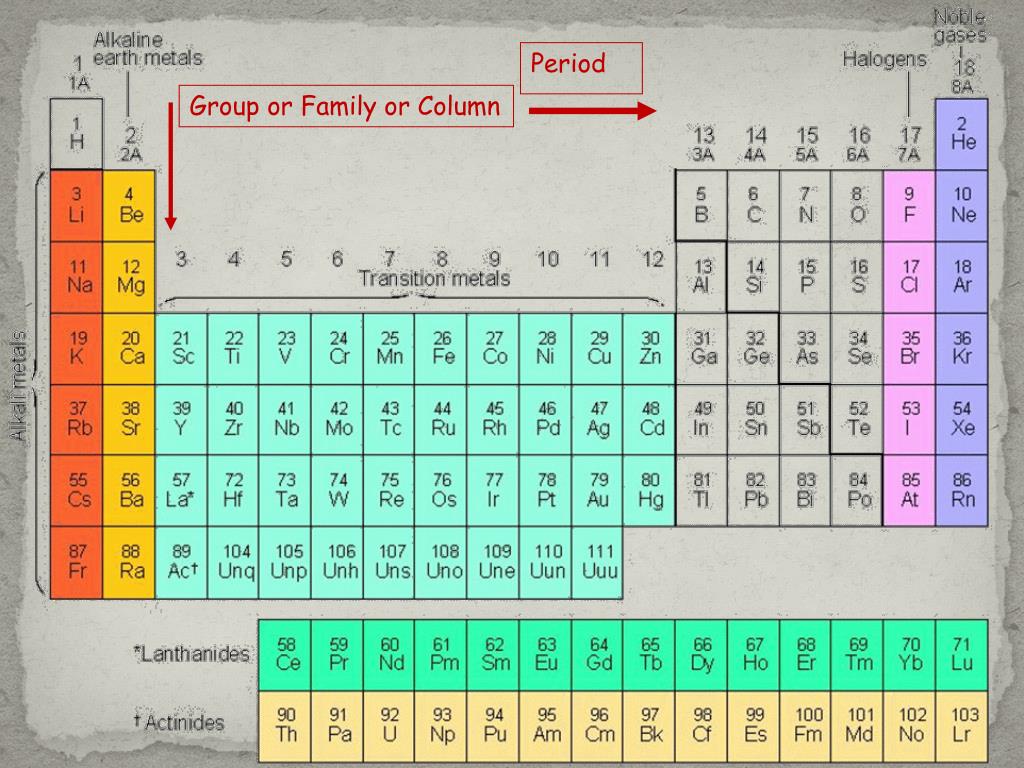
What is the name given to the group of elements that have the following valence shell electron configurations? a. What are the Group 18 elements called? 23. What are the Group 17 elements called? 22. What are the Group 2 elements called? 21. What are the Group 1 elements called? 20. GROUPS 3-12 f-ORBITALS BOTH Ca AND Sr HAVE TWO VALENCE ELECTRONS Ca GROUP 11 Cu, Ag, Au RADON (Rn) RADIUM (Ra) ONE ALKALI METALS 19. 
In going from top to bottom of any group, each element has more occupied energy level(s) than the element above it. What is the heaviest noble gas? What is the heaviest alkaline earth metal? 18. Would you expect strontium to be, chemically, more similar to calcium or rubidium and WHY? 16. In what type of orbitals are the actinide and lanthanide electrons found? 15. C and Pb PERIOD NUMBER = ENERGY LEVEL OF VALENCE ELECTRONSġ3. How does an element’s period number relate to the number of the energy level of its valence electrons? 6 2 7 5 Ne, NEON Ar, ARGON Kr, KRYPTON Th THORIUM 2 2 4 8 6 14 a. Which of the following pairs of elements belong to the same group? a. Which of the following pairs of elements belong to the same period? a. List, by number, both the period and group of each of these elements. Give the names and chemical symbols for the elements that correspond to these atomic numbers: a. State the number of valence electrons in an atom of: a. What is a group (also called a family)? How many are there in the periodic table? DMITRI MENDELEEV ATOMIC MASS ATOMIC NUMBER THE PROPERTIES OF THE ELEMENTS REPEAT PERIODICALLY A HORIZONTAL ROW IN THE PERIODIC TABLE 7 A VERTICAL COLUMN IN THE PERIODIC TABLE 18ħ. What is a period? How many are there in the periodic table? 6. By what property did Moseley suggest that the periodic table be arranged? 4. By what property did Mendeleev arrange the elements? 3. Who first published the classification of the elements that is the basis of our periodic table today? 2. 
Click Here for COPY Key Chemistry: The Periodic Table and Periodicity 1. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
